Objective
to compare the safety and efficacy of a bioresorbable occluder with a metallic occluder for transcatheter atrial septal defect (ASD) closure
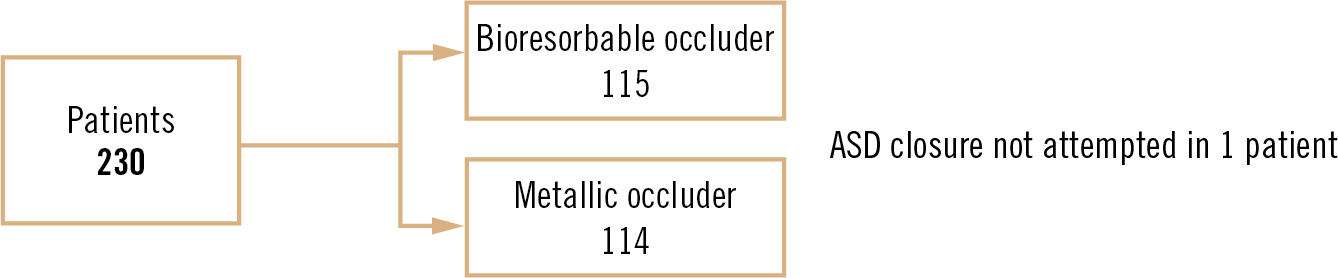
Study
multicentre, non-inferiority, open-label randomised clinical trial (1:1 ratio)
Population
patients aged 65, secundum ASD with the following anatomical criteria: 1. ASD diameter 5mm + right heart volume overload or left to right shunt with ASD diameter 28mm, 2. minimum distance 5mm from ASD edge to key intracardiac structures + minimum distance 7mm to atrioventricular valve, 3. no other indication for cardiac surgery
Endpoints
success rate of ASD closure at 6 months (defined as procedural success with residual shunt diameter 2mm)
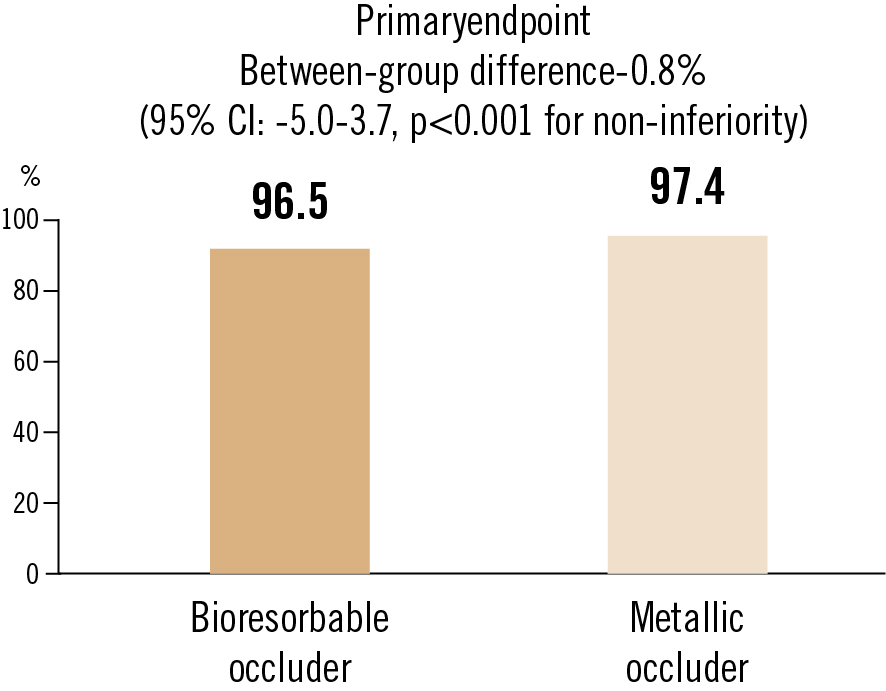
Conclusion
The bioresorbable occluder was found to be non-inferior to the metallic occluder for atrial septal defect closure, with degradation nearly complete by 2 years.
Ouyang et al. JAMA. 2025 Oct23.