Objective
to assess the safety and efficacy of a novel biodegradable patent foramen ovale closure device made of polydioxanone (PDO) monofilament when compared to conventional nitinol devices
Study
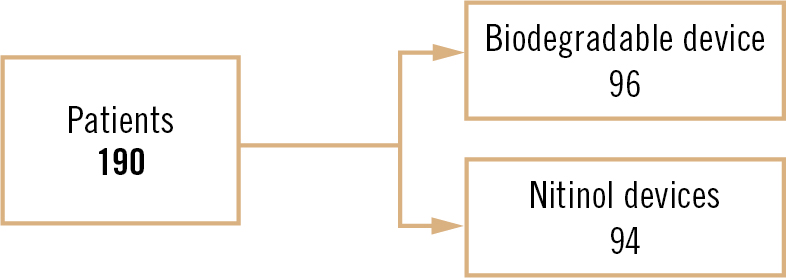
multi-centre, randomised controlled trial with non-inferiority design
Population
patients aged 18-65 years with a proven PFO on contrast echocardiography, at least one clinical indication for closure, and no contraindication to PFO closure
Endpoints
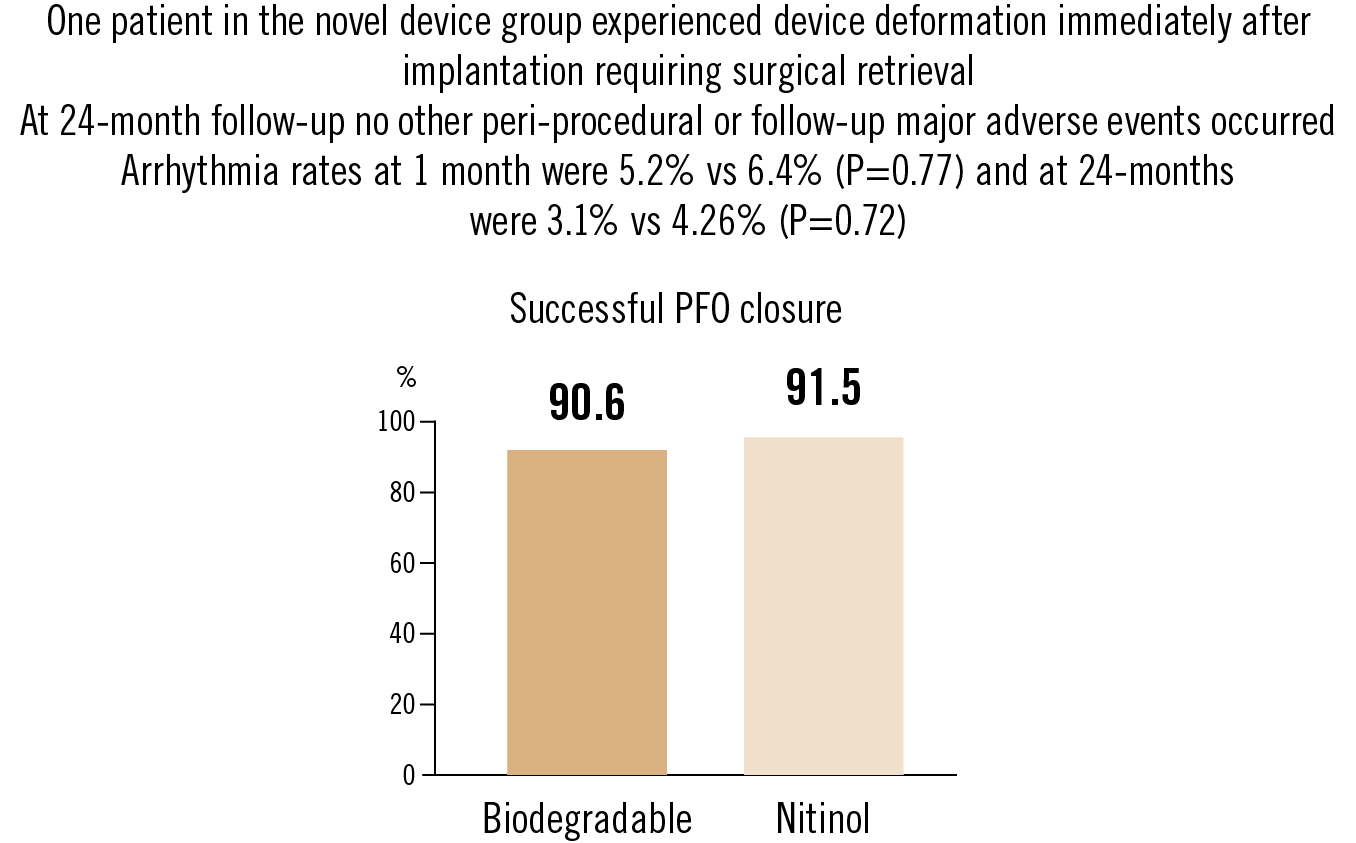
primary (efficacy): PFO closure success rate at 6 months, assessed with contrast echocardiography; secondary: major adverse events, arrhythmias and device degradation
Conclusion
A novel biodegradable PFO closure device was noninferior to the traditional nitinol devices for both efficacy and safety. 100% of echocardiographic evidence of the device had disappeared within 24 months of implantation.
Zhang et al. Circulation. 2026 Jan 13.